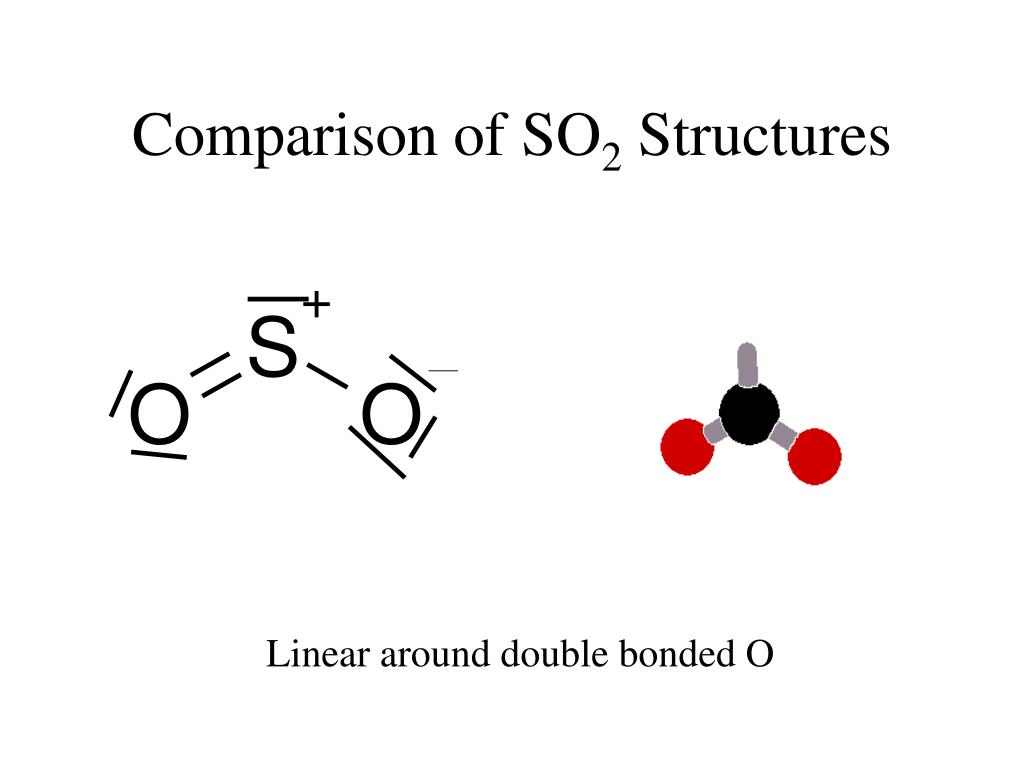
Sulfur hexafluoride (SF6) is a non-toxic gas that is used in a variety of applications due to its inert properties. Sulfur electronic configuration is 1s2 2s2 2p6 3s2 3p4 4. What is the biggest source of SO2?īurning fossil fuels is the largest source of SO2 in the atmosphere. The sulfur atom has one lone pair and each oxygen atom has two lone pairs. In the sulfur dioxide molecular geometry, there are two double bonds between the sulfur atom and oxygen atoms. How many lone pairs of electrons are present in the SO2 Lewis structure? If you have any questions, feel free to comment or send an email to 1. Some of the frequently asked questions are given below. SO2 gives a weak acid solution when dissolved in water.There are 5 lone pairs of electrons in the molecule of SO2.The sulfur dioxide molecule has two double bonds between the Sulfur atom and Oxygen atoms.The sulfur to oxygen ratio in sulfur dioxide is 1:2.The molecular geometry of sulfur dioxide is a bent shape.Both the sulfur and oxygen atoms have six valence electrons. The SO2 Lewis structure would consist of two oxygen (O) atoms and one sulfur atom.The carbon atom is in the central position as it is the least electronegative atom in the molecule. Two oxygen atoms are located on either side of the carbon atom.īoth oxygen atoms share electrons and form bonds with the central carbon atom. In this structure, there are two oxygen atoms and one carbon atom. CO2 Lewis StructureĪnother similar Lewis structure is the CO2 Lewis structure. These small particles may penetrate deeply into the lungs, and their sufficient quantity can contribute to health problems. SO x can react with other compounds in the atmosphere to form small particles. High emissions of sulfur dioxide in the air can lead to the formation of other sulfur oxides (SO x). Its high concentrations can cause inflammation and irritation of the respiratory system. It can irritate the skin and mucous membranes of the eyes, nose, throat, and lungs. Sulfur dioxide is a toxic gas and is directly harmful to human health. Related Links SiO 2 Lewis Structure Laughing Gas HCN Lewis Structure Sulfur Dioxide Effects on Humans This is another cause of the polarity of the sulfur dioxide molecule.Ĭheck the full article “SO2 is polar or nonpolar?”. 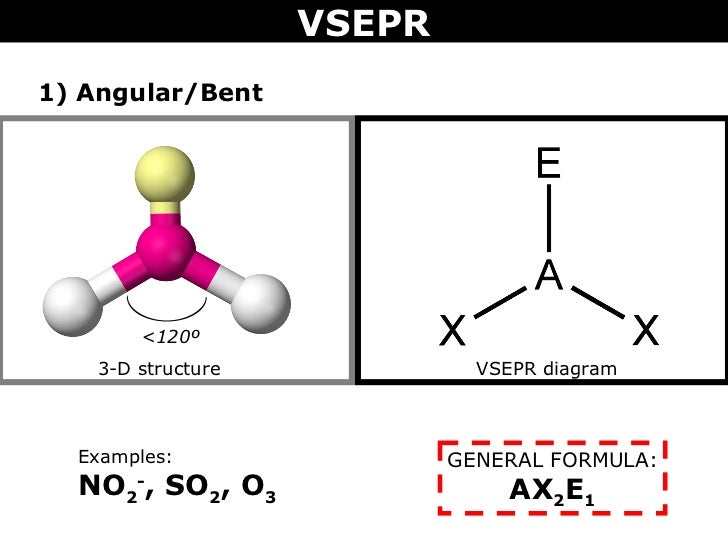 
In addition, the unbonded electrons on the sulfur and oxygen create repulsion between atoms. This makes SO2 a polar molecule like H 2S. Whereas the portion that has the sulfur atom has a slightly positive charge. The portion of the molecule that has both oxygen atoms on it is slightly negatively charged. Therefore, oxygen exerts more pull on the covalent bonds in sulfur dioxide. Oxygen has a greater electronegative potential than sulfur. The difference in electronegativity between sulfur and oxygen atoms creates polarity in the molecule. While reacting with nonmetals, both form covalent compounds, for instance, H 2 O, H 2S, CO 2, and CS 2.ĭissimilarities between oxygen and sulfur Oxygen. 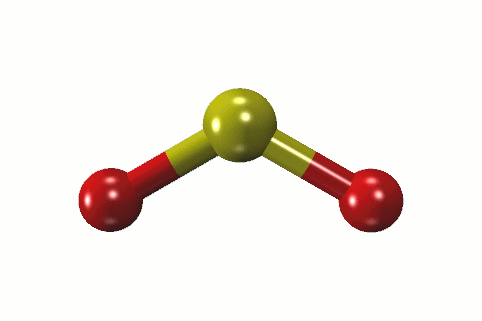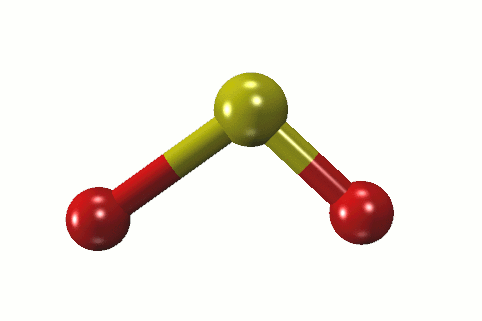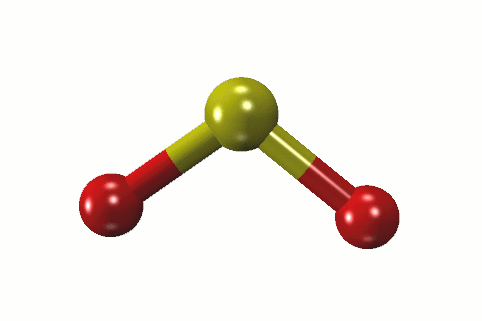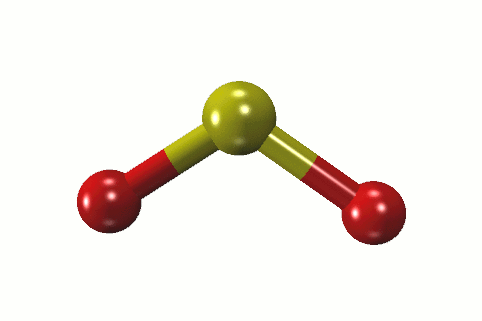
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
